- Research Summary
This research was conducted with support from Japan Agency for Medical Research and Development (AMED) through the joint participation of HGPI Chairman Kiyoshi Kurokawa as research and development representative and co-researchers HGPI Vice-Chairman Hiroaki Yoshida and Yoshiki Niimi, Assistant Professor of Fujita Health University, School of Medicine.
This research program aims to propose the best possible method for establishing an International Public-Private Partnership (PPP) that will promote dementia research in an effective and efficient manner and incorporate their methods into a concrete framework in Japan. It also aims to spread Japan’s knowledge so that international initiatives can be included in frameworks created abroad when they are proposed to related government agencies and international organizations.
Activities related to dementia measures undertaken by Health and Global Policy Institute
The Research Project on the Construction and Use of a Model for an International Public-Private Partnership (PPP) in Dementia Research
Research on projects and organizations related to dementia that make use of the PPP model in Japan and abroad
We conducted a survey of research projects and organizations related to dementia that make use of the PPP model. The survey also included representative organizations that are making progress in research on the formation of PPPs.
■Dementia research programs around the world-Large-scale, multi-institutional joint studies and partnerships
| Abbreviation | WW-ADNI | DIAN | A4 study |
|---|---|---|---|
| Official Name | World Wide Alzheimer's Disease Neuroimaging Initiative | Dominantly Inherited Alzheimer Network | Anti-Amyloid Treatment in Asymptomatic Alzheimer’s study |
| Year of Establishment | 2003 | 2008 | 2014 |
| Management | Alzheimer's Association | Washington University School of Medicine | Alzheimer's Therapeutic Research Institute |
| Collaborated organizations | 25 companies, 150 academic organizations, Foundation for the National Institutes of Health The National Institute on Aging | The National Institute on Aging | The National Institute on Ageing, Eli Lilly and Company, Philanthropic institutions |
| Aims | To help define the rate of progression of mild cognitive impairment and Alzheimer’s disease To develop improved methods for identifying the appropriate patient populations to participate in clinical trials. To standardize the methods used for conducting imaging scans and gathering and testing fluid samples | To find solutions to treat or To find solutions to treat or prevent Dominantly Inherited Alzheimer’s Disease (A rare form of Alzheimer’s), and potentially, all forms of Alzheimer’sprevent Dominantly Inherited Alzheimer’s Disease (A rare form of Alzheimer’s), and potentially, all forms of Alzheimer’s | To test whether a new investigational treatment, called solanezumab, an anti-amyloid antibody, can slow memory loss caused by Alzheimer’s disease |
■Dementia research programs around the world-Large-scale study-funding organizations
| Abbreviation | IMI | EPAD | DPUK | GAP | AMP | IADRP | GAAIN |
|---|---|---|---|---|---|---|---|
| Official Name | Innovative Medicines Initiative | European Prevention of Alzheimer Dementia Consortium | The MRC Dementias Platform UK | Global Alzheimer's Platform | Accelerating Medicine Partnership | International Alzheimer's and Related Dementias Research Portfolio | The Global Alzheimer’s Association Interactive Network |
| Year of Establishment | 2008 | 2015 | 2014 | 2013 | 2014 | 2010 | 2010 |
| Management | European Union、European Federation for Pharmaceutical Industries and Associations(EFPIA) | Dr. Serge Van der Geyten | Medical Research Council | The GAP Foundation | Foundation for the National Institutes of Health(FNIH) | NIH, National Institute on Aging and the Alzheimer’s Association | USC Laboratory of Neuro Imaging, USC Mark and Mary Stevens Neuroimaging and Informatics institute, Keck School of Medicine of USC |
| Collaborated organizations | Janssen Pharmaceutical, Eisai Pharma, Bayer Pharma, etc | 35 organisations including Eurpean Union, EFPIA companies and Universities, and research organisations | The National Institute on Aging (NIA), the National Institute of Bioimaging and Bioengineering (NIBIB), Araclon Biotech, Cambridge Cognition, MedImmune, the global biologics research & development arm of AstraZeneca, GlaxoSmithKline, Invicro, Ixico, Janssen Pharmaceuticals, in collaboration with Johnson & Johnson Innovation, SomaLogic. | Janssen, Eli Lilly and Company, Roche, Lundbeck | Food and Drug dministration(FDA), National Institutes of Health (NIH), AbbVie, Biogen, Bristol-Myers Squibb, GlaxoSmithKline, Johnson & Johnson, Eli Lily and Company, Merck, Pfizer, Sanofi, Takeda Pharmaceutical Company, Alzheimer‘s Association, Alzheimer’s Drug Discovery, American Diabetes Association, etc. | 40 organizations from 10 countries including, Administration on Ageing, CDC, Alzheimer’s Association, BrightFocus Foundation, Canadian Institute of Health Research, Alzheimer’s Research UK | 24 organizations including, Alzheimer’s Association, DIAN, Aibl, ADNI, I-ADNI, CAMD |
| Aims | To develop next generation vaccine, medicine, treatments | To improve the understanding of the early stages of Alzheimer’s disease and deliver new preventative treatments | To help define the rate of progression of mild cognitive impairment and Alzheimer’s disease To develop improved methods for identifying the appropriate patient populations to participate in clinical trials. To standardize the methods used for conducting imaging scans and gathering and testing fluid samples | To speed up the delivery of innovative therapies to those afflicted with Alzheimer’s by reducing the time and cost of Alzheimer’s disease (AD) clinical trials. | To transform the current model for developing new diagnostics and treatments by jointly identifying and validating promising biological targets for therapeutics | Enable public and private funders of Alzheimer’s research to coordinate research planning, leverage resources, avoid duplication of funding efforts and identify new opportunities in promising areas of growth. | Promotes big data sharing among a federated, global network of data partners who are studying Alzheimer’s disease and other dementias. To address the need to coordinate and leverage existing resources to advance research into the root causes of the disease, improve diagnostics and discover novel therapeutics, and find better ways to deliver care. |
■Dementia research programs in Japan – Large-scale, multi-institutional joint studies, partnerships, and large-scale study-funding organizations
| Abbreviation | J-ADNI | IROOP | ORANGE Registry |
|---|---|---|---|
| Official Name | Japanese-Alzheimer’s Disease Neuroimaging Initiative | Integrated Registry Of Orange Plan | Organized Registration for the Assessment of dementia on Nation-wide General consortium toward Effective treatment in Japan |
| Year of Establishment | 2007 | 2016 | 2015 |
| Management | University of Tokyo | National Center of Neurology and Psychiatry (NCNP), Japan Agency for Medical Research and Development | National Center for Geriatrics and Gerontology, National Center of Neurology and Psychiatry (NCNP), Tokyo dementia care research and training center, Ministry of Health, Labor and Welfare |
| Collaborated organizations | 30 academic organizations | National Center for Geriatrics and Gerontology, Yokohama Brain and Spine Center, Osaka City University Faculty of Medicine | 30 academic organizations |
| Aims | To conduct joint clinical studies with other facilities to develop criteria for evaluating the efficacy of AD drugs. To track healthy elderly patients, MCI patients, and early AD patients for 2 to 3 years. | To clarify symptoms that appear before the onset of dementia and to elucidate risk factors by improving lifestyles, habits, etc. To promote clinical research and clinical trials for the development of drugs expected to improve cognitive function. | To establish an appropriate care method for dementia by collecting information on people in various stages of health including: Healthy, Preclinical stage, MCI, Mild dementia, Moderate dementia, and Advanced dementia. |
This research identified that the essential element of an effective PPP is clarity in organizational values, mission, and goals.
Furthermore, those interviewed commented on the strong need for Japan to be involved in international research collaborations including those using the PPP model. To achieve this, it is important to strengthen the basic foundations of clinical trial research in Japan, including the practices by which participants and workers who can collaborate internationally are recruited. It is also crucial to secure stable funding streams to make all of this possible.
Collection of case studies on dementia research and research promotion efforts led by PPPs
In a study of fields that can be expected to be promoted by a PPP excluding drug discovery, we conducted interviews and other surveys with care service providers and related services, local governments, town planning experts, robotics experts, and IoT researchers. We also conducted a survey of precedent cases of research promoted by PPPs on topics other than dementia and used them as references for modeling the construction of a dementia PPP.
■Challenges facing stakeholders undertaking advanced efforts in areas related to dementia and their needs
| Stakeholder | Challenges and needs |
|---|---|
| People with dementia and their families |
|
| Medical and care service providers |
|
| Companies and academia involved in clinical trials and clinical research |
|
| Companies providing technology and services that promote and improve care |
|
| Self-governing bodies that implement progressive initiatives |
|
| As a whole |
|
■Suggestions for the PPP mechanism in Japan
| Classification | Suggestions for PPP mechanism |
|---|---|
| Visions, missions, and values |
|
| Business model |
|
| Administrative structure |
|
| Publicity campaigns |
|
| Collaboration with other organizations |
|
| Institutional design |
|
Interviews identified the need for a PPP to facilitate knowledge sharing within the research community and to assess new technology. Also, interviews highlighted the importance of having a clear shared vision and mission within the PPP and of promoting transparency and impartiality in project management.
Collaboration with the World Dementia Council (WDC)
Interviews with stakeholders revealed that they are highly interested in the role that Japan is expected to fill in the WDC and in other global policy planning endeavors. After sharing opinions with the WDC on this matter, the WDC decided to investigate the current situation of and future topics in each field it focuses on.
■Suggestions and expectations for Japan from the WDC
| Category | Suggestions and expectations for Japan |
|---|---|
| Finance |
|
| Integrated Drug Development |
|
| Research, Open Science, and Data |
|
| Care |
|
| Risk Reduction |
|
These conversations allowed for opinions on the concept of the PPP model to be exchanged and they reaffirmed the need for Japan to actively contribute to the WDC. They also stressed the importance of long-term strategic efforts for effective drug discovery and the standardization of care and evaluation methods.
Multinational comparative analysis of the current state of PPPs for dementia
A multinational comparative analysis was carried out to examine the current status of international PPPs for dementia research. It identified elements and functions required for cooperation between industry, government, and academia regarding dementia research in Japan.
- The number of people with dementia
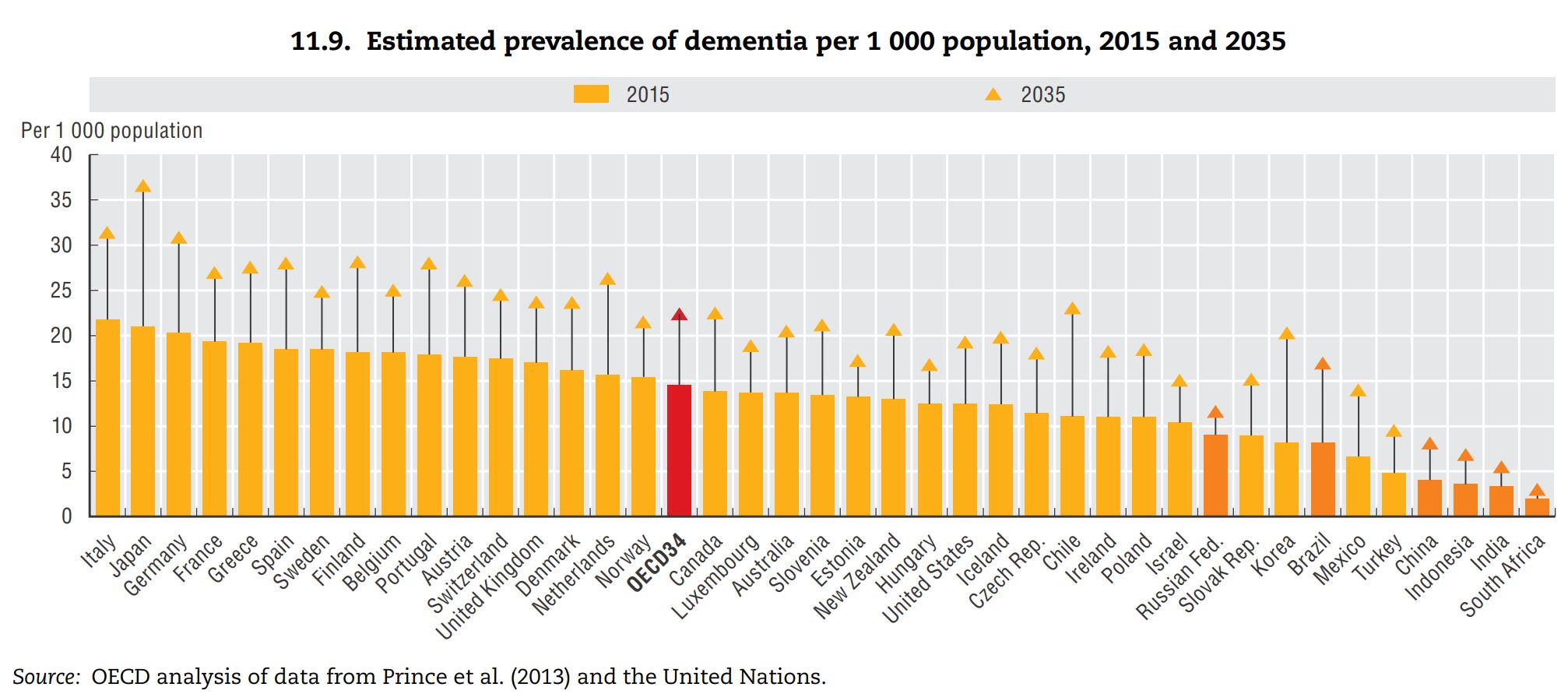
According to the estimate for the number of people with dementia in member nations of the OECD reported in the OECD’s Health at a Glance 2015, both the prevalence of dementia and its future rate of growth in Japan are high. Western Europe and the Nordic countries mostly had high levels of dementia among the OECD member states, and countries such as Chili, South Korea, and Brazil were projected to have high growth rates in the future.
- Dementia-related research and development funding
Although funding for the Government Budget Appropriations or Outlays on Research and Development (GBAORD) in Japan is around the same as countries excluding the United States and the United Kingdom, the amount of funding for dementia and neurodegenerative disease-related research and development is very low.
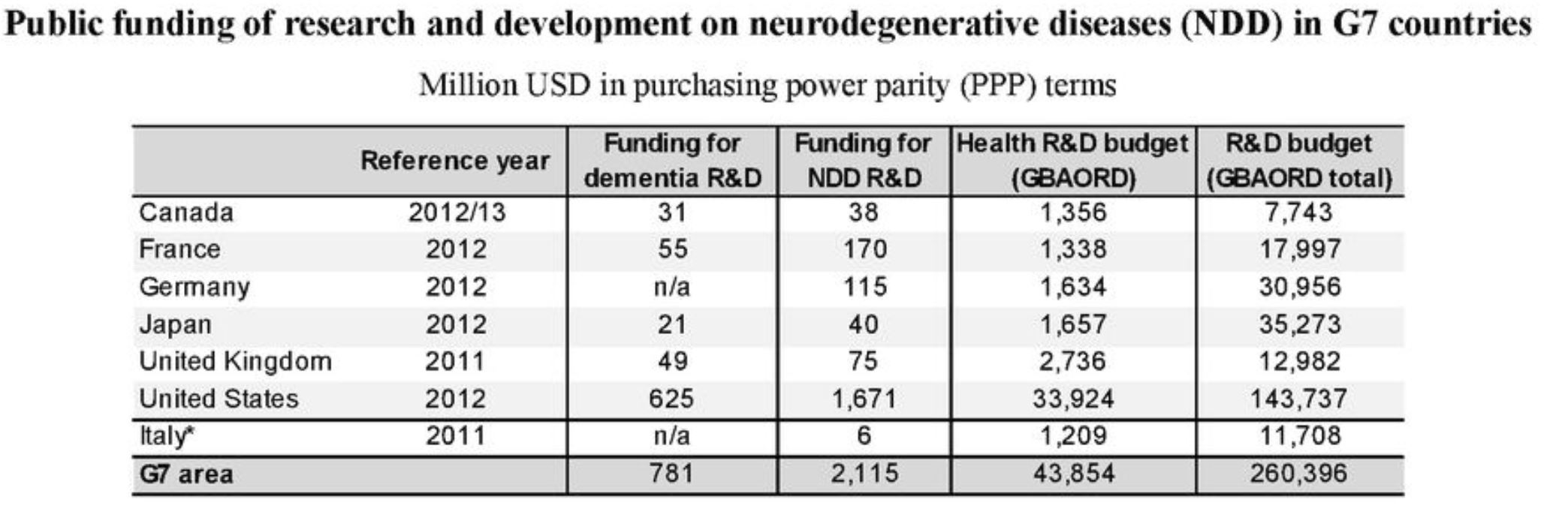
Source: OECD Health Policy Studies Addressing Dementia
- Cost of Dementia in Japan
The overall cost of dementia in Japan is 14.5 trillion yen (2014 estimation). The breakdown of costs reveals that the costs of nursing and informal care together account for a much larger proportion (87%) of total costs than medical treatment.
Regarding the dementia related cost ratio around the world, Asian countries have especially low medical costs, and Western countries often have high cost ratios for social centers.
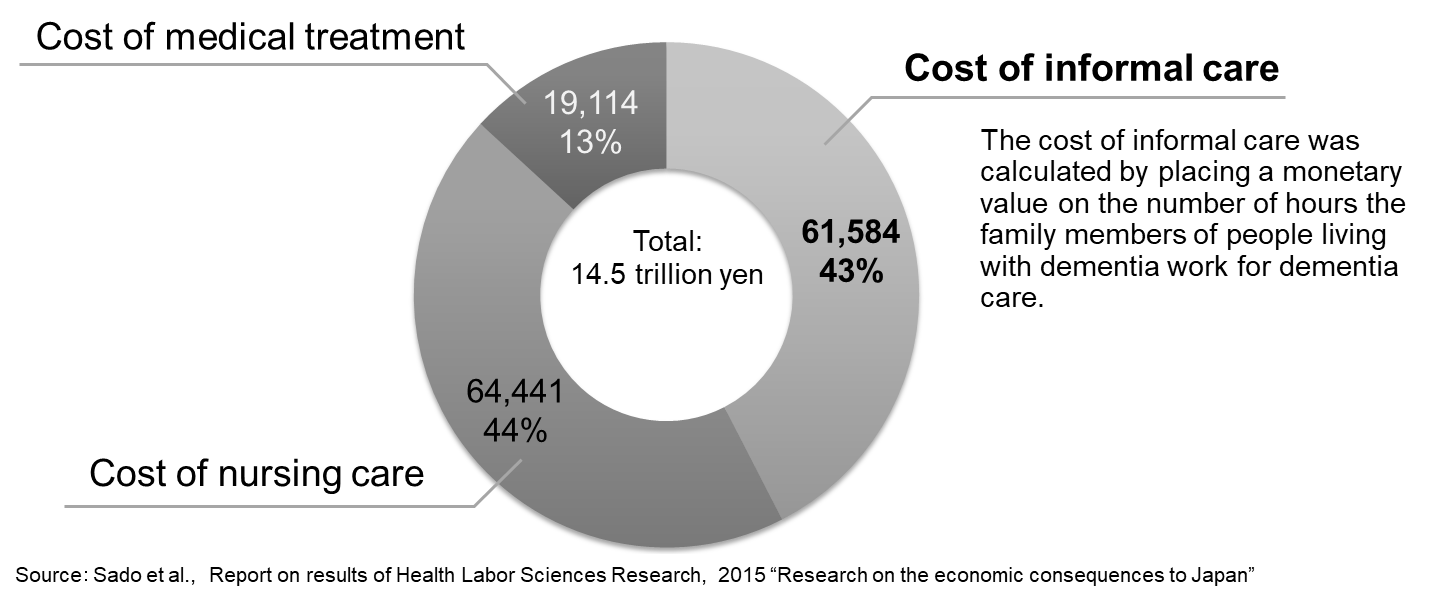
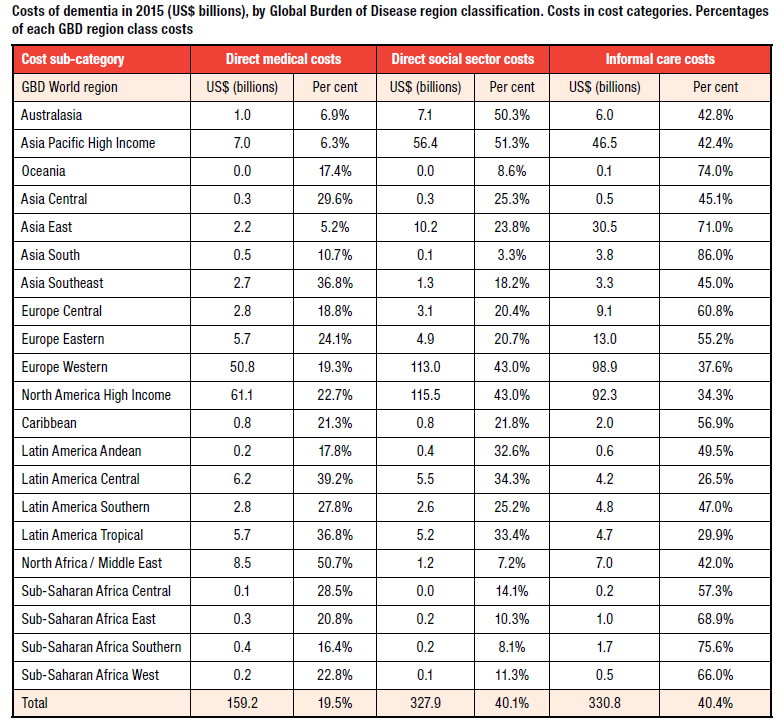
Source: World Alzheimer Report 2015
- Status Development status for national strategies and guidelines
The United States signed the National Alzheimer’s Project Act (NAPA) into law as a national project on Alzheimer’s disease, while Britain and Japan have implemented policy measures as part of their national strategies. It is necessary for Japan to consider establishing basic laws in the future like the United States. Furthermore, Japan’s guidelines on clinical trials are inadequate compared to Western countries, and therefore their improvement will be a goal for the future.
■Dementia-related national strategies and guidelines in Western countries and Japan
| United States | United Kingdom | Japan | ||
|---|---|---|---|---|
| Dementia related national strategies | The National Alzheimer’s Project Act (NAPA) | Living well with dementia: A National Dementia Strategy | Comprehensive Strategy to Accelerate Dementia Measures (New Orange Plan) |
|
| Guidance/ Guidelines | Name | Guidance for Industry, Alzheimer’s Disease : Developing Drugs for the Treatment of Early Stage Disease | Draft guideline on the clinical investigation of medicines for the treatment of Alzheimer’s disease and other dementias | Guideline on the clinical evaluation of medicines for the treatment of Alzheimer’s Disease (AD) |
| Explanation | The FDA’s current approach covers areas such as: diagnostic methods for early-stage Alzheimer’s disease patients who are participating in clinical trials; patient selection and diagnostic methods for patients for whom the disease has the possibility of progressing; directions for setting the endpoints of clinical trials; and directions for using biomarkers to measure treatment outcomes. | These guidelines aim to create new diagnostic criteria for AD, including early-stage and asymptomatic AD patients based on the results of impact assessments; select treatments based on outcomes for each stage of progress of AD; enable the effective use of biomarkers; and design trials to determine the long - term effectiveness and safety of dementia treatment drugs. | Currently being produced. | |
The results of this project emphasized the need for Japan to become involved in improving relevant guidance and guidelines for dementia. It also emphasized the importance of developing a registry framework for clinical trials in order to further advance global collaborations in dementia research.
Organizing meetings involving people with dementia and their families and representatives from government, academia, and the private sector
An advisory board and an expert committee that brought together people with dementia and their families and representatives from industry, government, and academia was established in order to exchange opinions regarding the promotion of research by a PPP. (*All affiliations are current at the time of writing)
- Expert Committee

- Advisory Board

Proposals for the establishment of an international PPP model and policy
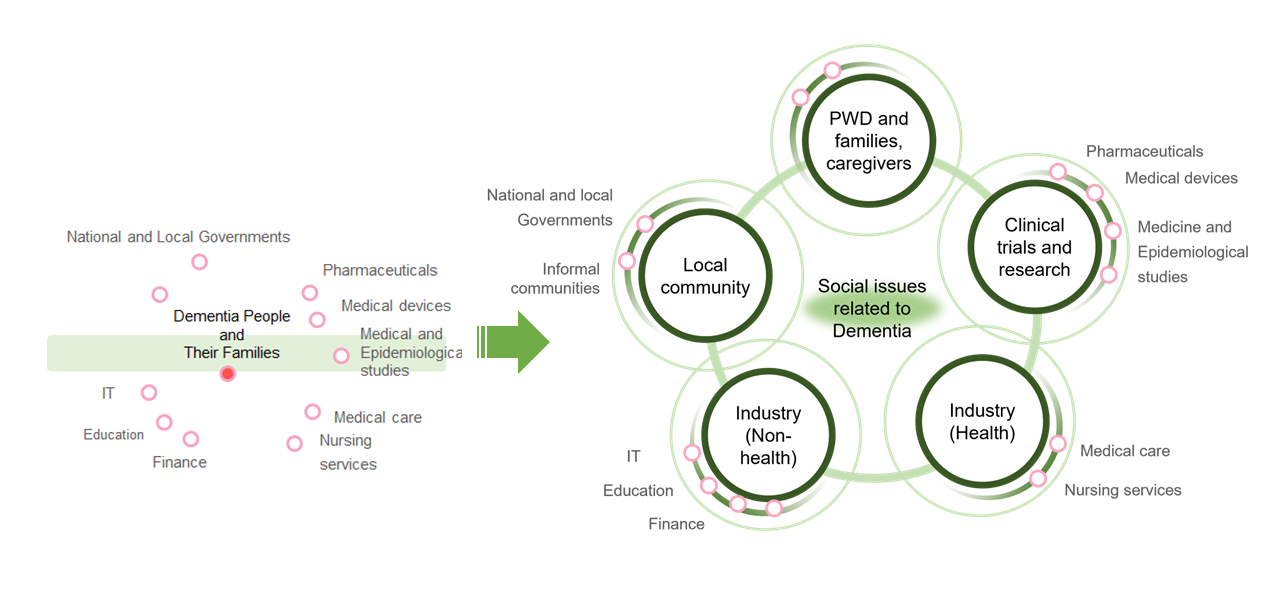
Three key approaches that will enable Japan to demonstrate leadership in Asia as a society experiencing advanced aging have been identified. First, the PPP must provide a platform for data collection and assessment that can facilitate discussion among organizations from various sectors. Second, the PPP must also function as a high-quality dementia registry that can enable Japan’s participation in large-scale international clinical trials. Last but not least, the PPP should function as an organization that can raise awareness of dementia among the public and thereby promote understanding and acceptance.
- Stakeholders for promoting PPP
The promotion of a PPP for dementia policy will require not only cooperation between stakeholders and people with dementia and their families, but also mutual cooperation among stakeholders. Furthermore, people with dementia should not be positioned to only receive unilateral support; rather, we should aim to create a world in which those with dementia can also actively contribute to tackling the challenges dementia presents to society.
- The ideal dementia PPP as suggested by this research
Detailed below is a vision for the field of dementia and for society that the dementia PPP structure proposed by this research aims to create. In the future, we must present a vision and mission and create a PPP roadmap.
-
- In pursuit of an effective and efficient PPP
Ideally, in the future this PPP will:- Develop research themes and activities based on the needs of people with dementia and their families.
- Appropriately prioritize research funding and activities based on an understanding of the issues surrounding dementia and evaluate the effects of research constantly.
- Enable all stakeholders to access and share data and knowledge from dementia research and related services.
- Encourage the private sector to invest actively in dementia policy and motivate further participation in this area.
- Actively adopt and utilize scientific and technical innovations for its work, including innovations related to ICT and robotics.
- In pursuit of an effective and efficient PPP
-
- Goals of the dementia PPP
The PPP should aim to:- Solve latent social issues in Japan that are at the backdrop of dementia-related problems, including but not limited to issues related to low birthrates and an aging population; issues that women face; and issues related to people leaving employment to care for family members. Multifaceted and systematic solutions should be developed.
- Take the initiative in creating global policy and serve as a hub for global research as a PPP based in the country with the highest proportion of elderly people in the population in the world. It should build links to the government’s Asia Health and Human Well-Being Initiative.
- Goals of the dementia PPP
- The functions that Japan’s PPP platform on dementia should have, and the course of action that it must take
We will establish a PPP platform on Dementia so that Japan can successfully manage the social challenges brought about by long life expectancies among Japanese people and to demonstrate leadership in Asia as a country already experiencing population aging.
This platform shall:
|
*The contents of this page is based on the information as of March 2017 when we published our research results.

